HDRP Newsletter, April 2023
Message from the Acting Associate Director

Since the last Healthcare Delivery Research Program (HDRP) newsletter in Fall 2022, there have been notable updates. Our esteemed Associate Director, Dr. Paul Jacobsen, retired at the end of December 2022. We are currently recruiting for his replacement.
The end of 2022 and the beginning of 2023 included several virtual workshops and webinars led by HDRP staff, and several more are planned for the coming year. We hope you will join us for these events, which are described below.
In this issue of the newsletter, we highlight research activities, HDRP events, and more. Additionally, you will find information about new funding opportunities and data resources. Please reach out to me or any of our staff to learn more about these activities and funding opportunities. A full staff listing is available on our HDRP Staff page.
This is an exciting time for healthcare delivery research. I look forward to working together to realize the HDRP mission to improve the delivery of cancer-related care and improve health outcomes for individuals and populations.
Sincerely yours,
Janet de Moor, PhD, MPH
HDRP Events: Upcoming and Recap
HDRP continues to lead efforts that highlight scientific priorities for healthcare delivery research. Visit our News & Events page for comprehensive updates under "All Events".
SEER-MHOS Webinar Series
The National Cancer Institute (NCI) supports a research resource linking Surveillance Epidemiology and End Results (SEER) cancer registry data with Medicare Health Outcomes Survey (MHOS) data from the Centers for Medicare & Medicaid Services. SEER-MHOS offers investigators the opportunity to examine cancer health outcomes, such as health-related quality of life and activities of daily living, along with clinical and epidemiologic information. This webinar series is intended for researchers at all levels interested in learning more about the SEER-MHOS data resource. The first webinar on propensity and health scoring data took place on March 14, 2023. Scheduled dates for webinars 2 and 3 are confirmed for June 20, 2023 and September 12, 2023, respectively.
Healthcare Teams 2023-2024 Cyber Discussion Series
The goal of this webinar series is to discuss lessons learned by researchers and cancer care delivery teams about how to continue cancer care and research operations across the cancer continuum, and how to work effectively with clinical and community organizations, patients, and their families during some of our nation's most catastrophic events. Care coordination needs among populations that experience cancer disparities (racial/ethnic minority, under-resourced and historically disadvantaged communities) and populations disproportionately affected by public health emergencies will be a focus.
- On May 4, 2023, HDRP is hosting Session 1 the 2023-2024 Healthcare Teams (HCT) Cyber Discussion Series on Teaming, coordination challenges, and approaches to cancer care during disasters and public health emergencies. This interactive session will discuss teaming and coordination challenges for clinical care and research operations during disasters and public health emergencies. This session is part of a larger series that will discuss lessons learned by researchers and cancer care delivery teams about how to continue care across the cancer care continuum.
- Session 2: June 30, 2024, "Research examining disaster impact and resilience in cancer care delivery"
- Session 3: January 2024, "Global perspectives on cancer care teaming and resilience during natural and public health emergencies"
- Session 4: June 2024, "Patient and caregiver experiences navigating cancer during emergencies"
Measures and Methods to Advance the Science of Teaming and Coordination in Cancer Care
On September 28, 2023, HDRP will host a virtual meeting with the goal of strengthening the robustness of care delivery research on teamwork and care coordination constructs. Long-term outcomes include identifying instruments that may be good candidates for common data elements or gold standard measures, pooling of data across studies, and greater impact on the practice of teamwork and clinical outcomes. Registration information will be available soon.
A recap of recent HDRP-led webinars and workshops are listed below.
October 2022
- On October 6, 2022, HDRP held meeting on Challenges and Opportunities for Addressing Financial Hardship. The NCI awarded administrative supplements to eleven NCI-designated Cancer Centers to conduct research to develop or expand their capacity and infrastructure to deliver services to cancer patients and their families. This meeting convened investigators to share findings from their studies and stimulate conversation about the challenges and opportunities of this type of research.
- On October 27, 2022, the Addressing Social Risks in Cancer Care Delivery Webinar Series rounded out its schedule with a discussion about active treatment, survivorship, and challenges faced by a person diagnosed with cancer (from treatment to follow-up care). Panelists used a case study to facilitate discussion for this interactive session on team-based care approaches for integrating social care into cancer care for newly diagnosed patients.
January 2023
- On January 26, 2023, HDRP hosted a pre-application webinar for the reissuance of Research on Interprofessional Teamwork and Coordination During Cancer Diagnosis and Treatment (NOT-CA-22-014), which addressed care coordination challenges to encourage research to improve interprofessional teamwork, address care coordination challenges within care settings, and advance knowledge about how to design and carry out team-based care to improve quality and patient outcomes.
March 2023
- On March 14, 2023, HDRP held its first webinar of the SEER-MHOS Webinar Series on Propensity Scoring and Health Outcomes Data: Comparing Cancer and Non-Cancer Populations in the SEER-MHOS Data Resource to provide an overview and methods for this cancer health outcomes data resource.
Research Spotlights
A special series on teams in cancer care was published in JCO® Oncology Practice (JCO OP) – an American Society of Clinical Oncology Journal (ASCO) - on January 1, 2023 (volume 19, number 1) that included 15 peer-reviewed papers on the topic of team-based cancer care. This special series highlights a long-standing NCI-ASCO dialogue, led by Veronica Chollette, RN, MS and Sallie Weaver, PhD, MHS, to strengthen the science of teaming and adoption of evidence-based team-based care in clinical practice. The series builds on an NCI-ASCO partnership begun in 2014 that culminated in the first special series on teams in cancer care published in 2016
was published in JCO® Oncology Practice (JCO OP) – an American Society of Clinical Oncology Journal (ASCO) - on January 1, 2023 (volume 19, number 1) that included 15 peer-reviewed papers on the topic of team-based cancer care. This special series highlights a long-standing NCI-ASCO dialogue, led by Veronica Chollette, RN, MS and Sallie Weaver, PhD, MHS, to strengthen the science of teaming and adoption of evidence-based team-based care in clinical practice. The series builds on an NCI-ASCO partnership begun in 2014 that culminated in the first special series on teams in cancer care published in 2016 . Collaborating to publish a second special series - initiated by the then JCO OP editor-in-chief and ASCO leaders Drs. John Cox and Michael Kosty - acknowledges opportunities for federal-private partnership and dialogue to sustain the focus on healthcare team effectiveness and team-based care in cancer care delivery.
. Collaborating to publish a second special series - initiated by the then JCO OP editor-in-chief and ASCO leaders Drs. John Cox and Michael Kosty - acknowledges opportunities for federal-private partnership and dialogue to sustain the focus on healthcare team effectiveness and team-based care in cancer care delivery.
The 2023 special issue highlights care delivery approaches, interventions, and quality improvement activities organized around four aspects of team-based care: organizing and staffing teams of teams, equity in teams, increasing involvement and specialization of Advance Practice Providers, and teams and technology. At a recent National Cancer Advisory Board, Dr. Monica Bertagnolli (NCI Director) highlighted their editorial contribution and urged the scientific community to read this special series to understand the “dynamics of cancer care delivery at all levels…for successful clinical research…for the implementation of new advances in cancer care and prevention.” In March 2023, both Veronica and Sallie have been rated among the top 5 reviewers by ASCO for the completion, timeliness, and quality of their review in this 2023 special issue.
Below is a list of recent publications related to this area of research:
- Toward Team-Based Care in the United States: 6 Years Later, JCO Oncology Practice (vol. 19, issue 1)
- Teamwork competencies for interprofessional cancer care in multiteam systems: A narrative synthesis, Journal of Interprofessional Change (vol. 36, issue 4)
- Team-Based Care for Cancer Survivors with Comorbidities: A Systematic Review, Journal for Healthcare Quality (vol. 44, issue 5)
PRO-CTCAE® Measurement System (Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events). The NCI's PRO-CTCAE Measurement System is a patient-reported outcome measurement system developed to capture symptomatic adverse events in patients on cancer clinical trials. It was developed to complement clinician-graded CTCAE standard toxicity reporting to assess symptomatic adverse events reported by patients and to improve the precision and reproducibility of adverse event reporting. PRO-CTCAE has become the standard for patient-reported toxicity in cancer clinical trials around the world. There are 1760 studies currently registered to use PRO-CTCAE and over 150 citations in the scientific literature, including 48 clinical trial citations (with treatment trials of new therapies and early phase trials). PRO-CTCAE has been translated and linguistically validated in 30 languages. Examples of this measurement system's impact are that it:
- Provides confirmatory evidence that a treatment regimen is tolerable,
- Provides evidence of a more favorable tolerability profile for one regimen, when two regimens of comparable efficacy are compared, and
- Can help inform treatment selection and can target vulnerable patients who need additional supportive care.
Below is a list of recent publications reporting data from PRO-CTCAE®:
- Clinician and patient reporting of symptomatic adverse events in cancer clinical trials: Using CTCAE and PRO-CTCAE® to provide two distinct and complementary perspectives, Patient Related Outcome Measures (volume 13)
- Patient self-reporting of tolerability using PRO-CTCAE in a randomized double-blind, placebo-controlled phase II trial comparing gemcitabine in combination with adavosertib or placebo in patients with platinum resistant or refractory epithelial ovarian carcinoma, Society of Gynecologic Oncology (volume 167, issue 3)
- Reliability and validity of PRO-CTCAE® daily reporting with a 24-hour recall period, Quality of Life Research (Published online: March 10, 2023)
HDRP Fiscal Year (FY) 2022 Grant Portfolio Updates
In FY 2022, NCI funded 62 new grants totaling over $32 million, which are held by HDRP. These grants supported research across the cancer continuum to improve cancer prevention, detection, diagnosis, treatment, and survivorship care.
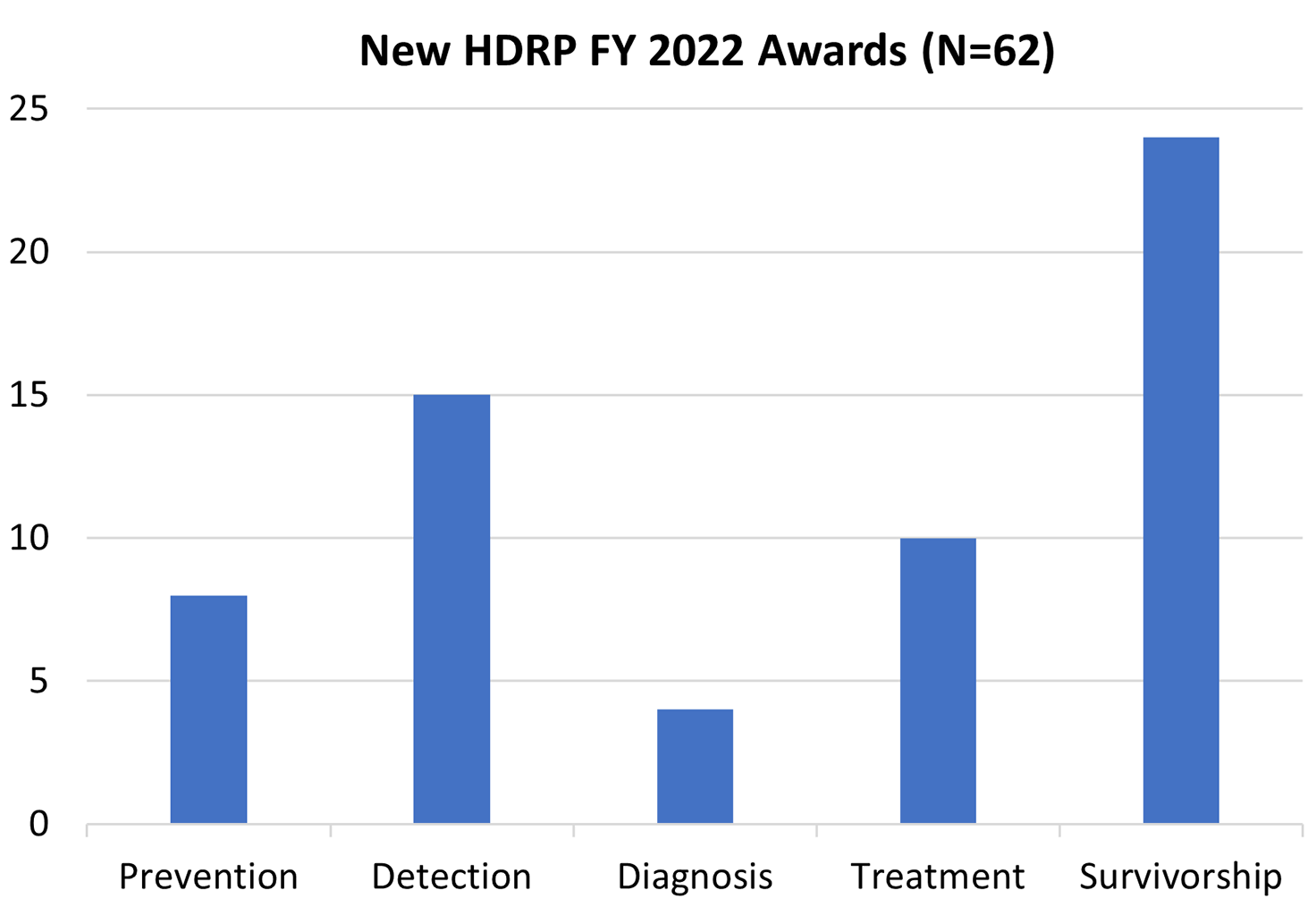
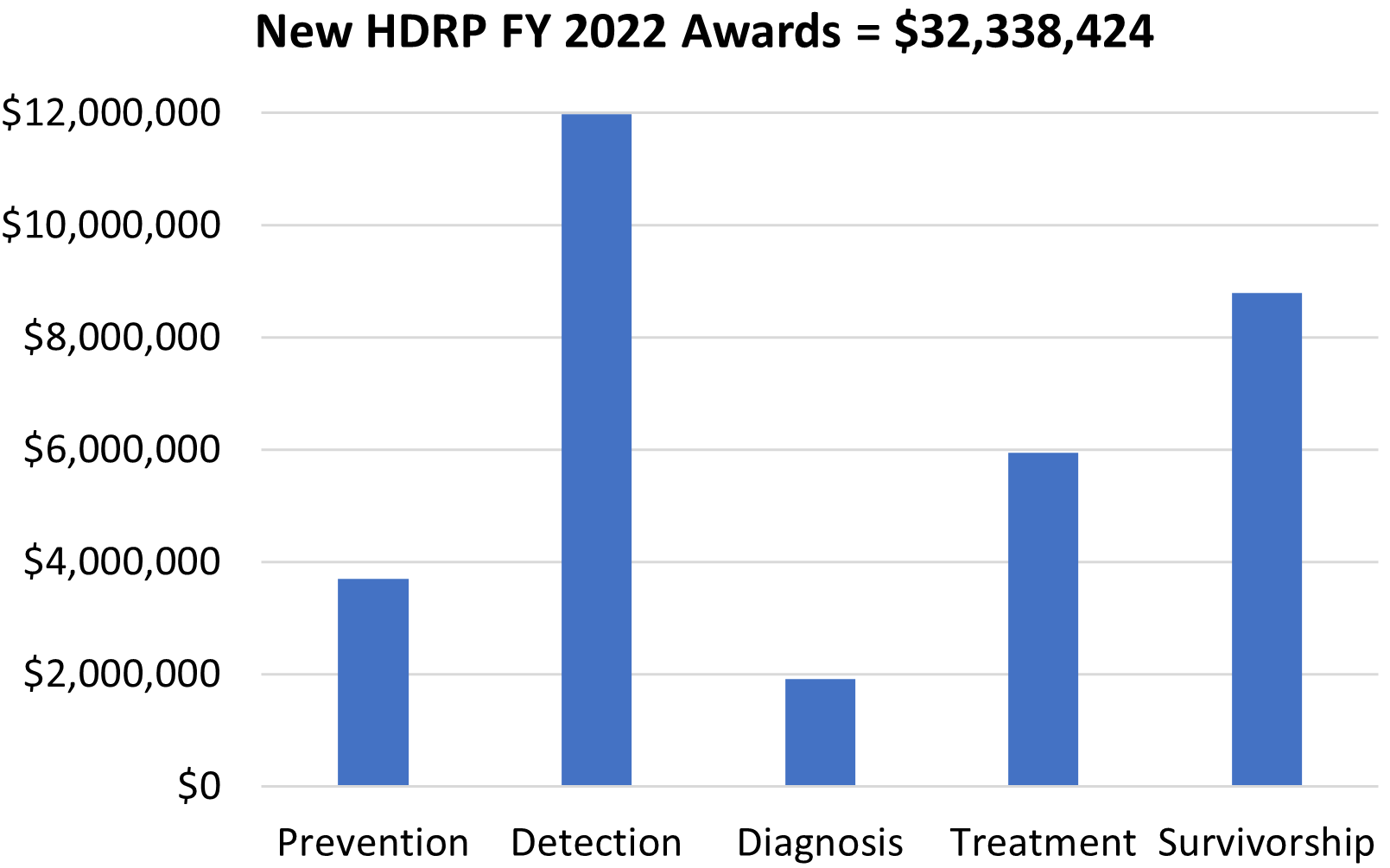
For a complete listing of all grants currently funded by HDRP, visit our funded grants page .
.
Policy Updates
Grants-related Information
Funding Policy Updates
The NCI has received its budget for FY2023. Visit the Division of Extramural Activities website for information on the Institute's budget, funding strategy, and more. The table below provides a snapshot of different funding mechanisms and associated paylines.
website for information on the Institute's budget, funding strategy, and more. The table below provides a snapshot of different funding mechanisms and associated paylines.
| Mechanism | FY2023 Funding Policy |
|---|---|
| Experienced and New Investigator R01 | 12th Percentile |
| Early-stage Investigator R01 | 17th Percentile |
| Exploratory/Developmental (R21) | 9th Percentile |
| Small Grant (R03) | 25 Impact Score |
AREA Grant (R15) Grant (R15) |
25 Impact Score |
Also, visit the NIH Grants & Funding page for more information on Standard Application Due Dates .
.
FOAs or NOFOs?
In early March 2023, the NIH Office of Extramural Research's Extramural Nexus notified the scientific research community of an important update. Following the footsteps of other federal agencies, funding opportunity announcements, or FOAs, are now being referred to as notice of funding opportunities, or NOFOs. The goal is to standardize terminology across the government of available grant support through funding opportunities. HDRP will be making the appropriate changes to various areas of our website.
notified the scientific research community of an important update. Following the footsteps of other federal agencies, funding opportunity announcements, or FOAs, are now being referred to as notice of funding opportunities, or NOFOs. The goal is to standardize terminology across the government of available grant support through funding opportunities. HDRP will be making the appropriate changes to various areas of our website.
NIH Expanding Usage of Notices of Special Interest (NOT-OD-19-107)
This notice informs the extramural community that the NIH is expanding and formalizing the use of Notices of Special Interest (NOSI) posted in the NIH Guide for Grants and Contracts to announce interest in specific scientific research topics.
How Does a NOSI Work?
A NOSI is a standard, formal format for NIH institutes to share and update their research priorities. A NOSI describes specific topics of interest and will direct applicants to one or more active notice of funding opportunitiess for application submission. A NOSI is not a NOFO and is listed as a notice in the NIH Guide for Grants and Contracts . Applicants should read NOSIs carefully for any special requirements related to that specific announcement.
. Applicants should read NOSIs carefully for any special requirements related to that specific announcement.
For more information about:
- The NIH expanding usage of NOSIs, visit the Notice (NOT-OD-19-107) page directly.
- NOSIs sponsored by HDRP, visit our funding opportunities page and read more in our Funding Opportunities section of this newsletter.
NIH Policy for Data Management and Sharing (DMS)
On January 25, 2023, the final NIH DMS Policy came into effect for any NIH-funded research that will generate scientific data, regardless of the research grant budget size. HDRP has compiled information from NIH on preparing DMS plans, research subject to the new NIH DMS Policy, and requesting and justifying costs for DMS.
For more information on this policy, including how to prepare DMS plans, associated costs, and more, visit Data and Resource Management and Sharing Plans on our Grantsmanship Resources page.
Announcements
Cancer Grand Challenges
On March 8, 2023, the NCI and Cancer Research UK announced nine new research challenges aimed at tackling some of the most profound problems in cancer research. This effort promotes the global research funding opportunity as part of the Cancer Grand Challenges Program . Healthcare delivery research will be important for tackling several of the challenges. Learn more about these challenges in the NCI Press Release
. Healthcare delivery research will be important for tackling several of the challenges. Learn more about these challenges in the NCI Press Release .
.
HDRP Funding Opportunities
This section includes links to new, highlighted NOFOs on healthcare delivery research published since the last edition of this newsletter. For a complete list of NOFOs, visit our funding opportunities webpage.
| Announcement Title | Announcement Number | Expiration Date |
|---|---|---|
| NOSI: Research on Oral Anticancer Agents in the Contexts of Utilization, Adherence, and Health Care Delivery | NOT-CA-20-026 | May 8, 2023 |
| NOSI: Research to Improve the Interpretation of Patient-Reported Outcomes at the Individual Patient Level for Use in Clinical Practice | NOT-OD-20-079 | May 8, 2023 |
| Addressing the Primary Care Needs of Cancer Survivors (U01 Clinical Trial Required) | RFA-CA-23-025 | July 1, 2023 |
| Advancing Research to Develop Improved Measures and Methods for Understanding Multimorbidity (R01 Clinical Trial Optional) | PAR-20-179 | September 8, 2023 |
| Identifying Innovative Mechanisms or Interventions that Target Multimorbidity and Its Consequences (R01 Clinical Trial Optional) | PAR-20-180 | September 8, 2023 |
Data & Other Resources
Updated SEER-CAHPS Data Available!
SEER-CAHPS is a publicly accessible data resource developed to enable investigators to conduct research on patient-reported experiences and quality of cancer care. These data provide a rich opportunity for analyses of Medicare beneficiaries’ experiences with their care at various stages of the cancer care continuum.
The SEER-CAHPS team is excited to share that we have recently updated the years of data available in this data resource. SEER-CAHPS now includes the following years of data:
- SEER Cancer Registry Data Through 2019
- CAHPS Experience of Care Survey Data Through 2019
- Medicare Claims Data Through 2019
We would also like to share that the state of Texas recently joined the SEER program. Hence, the cancer registry data now contains data from Texas residents from 2000-2019.
Updated SEER-Medicare Data Available!
SEER-Medicare data reflects the linkage of two large population-based sources of data that provide detailed information about Medicare beneficiaries with cancer.
The SEER-Medicare team is excited to share that we have recently updated the years of data in this data resource to include cancer cases diagnosed in 2018-2019 and Medicare data from 2019-2020.
For additional information on SEER-linked data and other tools and resources, visit our Data & Tools page.
HDRP News You Can Use!
Employment Opportunities
Cancer Research Training Award (CRTA): Healthcare Assessment Research Branch (HARB)
HARB promotes population-based research on demographic, social, economic, and health system factors as they relate to providing cancer-related screening, treatment, and survivorship services. The branch welcomes interested candidates for this post-doctoral fellowship.
Training Opportunities
Please visit the training section of our website to learn more about HDRP-specific and NIH/NCI-relevant opportunities.
