HDRP Newsletter, March 2026
Message from the Associate Director

On behalf of my colleagues in the Healthcare Delivery Research Program (HDRP), I hope that 2026 is off to a good start. As we begin the year, there are two important changes I would like to highlight.
First, this year marks an important leadership transition within the Division of Cancer Control and Population Sciences (DCCPS), of which HDRP is one of four research programs. In February, we bid farewell to Dr. Katrina Goddard, who retired from federal service after providing exceptional leadership as DCCPS Director. We are deeply grateful for her vision and commitment to advancing cancer control and population sciences and are pleased to welcome our Deputy Director Dr. Gary Ellison
(DCCPS), of which HDRP is one of four research programs. In February, we bid farewell to Dr. Katrina Goddard, who retired from federal service after providing exceptional leadership as DCCPS Director. We are deeply grateful for her vision and commitment to advancing cancer control and population sciences and are pleased to welcome our Deputy Director Dr. Gary Ellison as acting director of DCCPS. Dr. Ellison brings tremendous experience and thoughtful leadership to this role, and we look forward to working with him to continue advancing the Division’s mission.
as acting director of DCCPS. Dr. Ellison brings tremendous experience and thoughtful leadership to this role, and we look forward to working with him to continue advancing the Division’s mission.
Second, NIH is implementing a new funding strategy that moves beyond a strict payline model. We encourage you to learn more about these changes in the NIH Extramural Nexus article on implementing a unified NIH funding strategy . As always, we encourage investigators to stay in close communication with their Program Directors to better understand how evolving policies may apply to their work.
. As always, we encourage investigators to stay in close communication with their Program Directors to better understand how evolving policies may apply to their work.
In this newsletter, we have compiled resources, guidance, and updates to keep you informed about developments at NIH and within HDRP. I hope this information will support the important work that you are doing. Please feel free to reach out to me or any member of our staff to learn more about HDRP activities and available funding opportunities and data resources for healthcare delivery research.
Sincerely yours,
Janet de Moor, PhD, MPH
HDRP Events: Recap
As 2026 events are scheduled, they will be posted to our News & Events page. A recap of HDRP-led webinars and workshops since our last newsletter are listed below. To view post-meeting materials, visit the HDRP website and relevant event links.
Archives for the following sessions are available upon request. Please contact us at NCIHDRP@mail.nih.gov with the date and title of any session of interest to be furnished with a link to the recording.
Recap
SEER-MHOS Webinar Series
NCI supports a research resource linking Surveillance Epidemiology and End Results (SEER) cancer registry data with Medicare Health Outcomes Survey (MHOS) data from the Centers for Medicare & Medicaid Services. SEER-MHOS offers investigators the opportunity to examine cancer health outcomes, such as health-related quality of life and activities of daily living, along with clinical and epidemiological information.
- On January 15, 2026, HDRP hosted the second of three webinars in a series entitled, SEER-MHOS 2026 Update: Your Guide to the New Data Linkage. This webinar introduced the 2026 SEER-MHOS data linkage, with a focus on new data sources now available to the research community and explored uses of the newly linked Medicare Advantage Encounter data and the range of novel opportunities these additions present. Additionally, it described the available self-reported symptom variables, including sleep disturbance and cognitive function, that have now accumulated enough years of data to support many future research projects. For part one, visit: Measuring Prescription Opioid Use Using Medicare Part D Claims Data in SEER-MHOS Research Projects.
Healthcare Teams (HCT) Cyber Discussion Series
The goal of the HCT initiative is to advance a program of research supporting the development and implementation of evidence-based strategies that improve healthcare team functioning, care coordination, and cancer-related outcomes.
Advancing Team-Based Care Delivery Models & Interventions Across the Cancer Continuum
On January 16, 2026, HDRP hosted Advancing Team-based Care Delivery Models & Interventions in Treatment and Survivorship, the second of a two-part webinar series that examined multidisciplinary, interprofessional team-based care delivery models and related interventions to improve care coordination and address care quality and timeliness at multiple points along the cancer continuum. This session (1) discussed survivor and caregiver insights on care coordination challenges and burden, (2) highlighted studies examining team-based interventions addressing treatment and survivorship, (3) discussed strategies to address challenges in coordination and teaming, and (4) discussed challenges and strategies for effectively translating research findings into clinical practice. For part one, visit: Advancing Team-Based Care Delivery Models and Interventions in Cancer Screening and Risk Assessment in Primary Care.
Advancing Cancer Care Research in Medicare Advantage: Examining the Issues, Data, Measures, and Methods Series
Dramatic increases in Medicare Advantage (MA) enrollment over the past decade have heightened the need to examine cancer-related care quality and access within the MA program. This webinar series is designed to provide researchers with state-of-the-art information regarding (1) available data to capture the structures of MA plans and care delivery in MA; (2) challenges and best practices to using these data; and (3) analytic approaches used to study how MA enrollment affects cancer-related care.
- On August 5, 2026, HDRP hosted Using data from the Centers for Medicare & Medicaid Services (CMS) to measure cancer-related care and outcomes in Medicare Advantage (MA), the first in a three part series, provided an overview of data resources available to study cancer-related care, described the challenges in measuring cancer care in MA, and propose potential strategies for data users.
- On August 7, 2026, HDRP hosted Measurement and assessment of Medicare Advantage (MA) plan types, benefits, networks, the second in a three-part series, identified potential mechanisms through which plan characteristics may affect outcomes and describe key data sources available to describe plan characteristics and key measures.
- On August 14, 2026, HDRP hosted Medicare Advantage enrollment and plan selection behaviors, and causal inference methods to address selection, the third in a three-part series, examined current patterns of enrollment and plan switching among cancer survivors and what is known about relevant patterns. The webinar also discussed potential study designs that support causal inference in a complex and dynamic choice environment.
Data & Other Resources
HDRP supports the development and maintenance of a variety of research data and tools. These resources can be used to address research questions at various points across the cancer control continuum. Below, we highlight a subset of the data resources available to the extramural research community.
SEER-linked Data Updates
SEER-MHOS links NCI’s SEER cancer registry data with the Centers for Medicare and Medicaid’s Medicare Health Outcomes Survey (MHOS). SEER-MHOS is a data resource that allows researchers to examine health and quality of life among Medicare Advantage beneficiaries before and after a cancer diagnosis. Below are some updates:
- SEER-MHOS has been updated. The linkage now extends SEER data for persons diagnosed up to 2021, Medicare enrollment up to 2022, and survey data from 1998-2023.
- New Data Source available via SEER-MHOS! The data resource now includes Medicare Advantage encounter data. Researchers can conduct analyses that combine patient-reported outcomes with information on healthcare utilization and clinical events among Medicare Advantage beneficiaries from 2015 onwards. Access this link for more information on what is covered in the Medicare Advantage encounter data.
- With the inclusion of the Illinois SEER registry, SEER-MHOS now includes data from 22 SEER registries, covering approximately 46% of the U.S. population.
- Several patient-reported outcome domains added to SEER-MHOS in 2013 now can be pooled to achieve sample sizes to support analyses. Domains include pain interference, sleep disturbance, and cognitive function. For more information on patient-reported outcomes available in the MHOS survey data, visit this link.
To learn more about other data resources supported by HDRP, visit our data and Other Resources page.
Research Spotlight
New Publication from NCI/AcademyHealth Visiting Scholar Dr. Virginia LeBaron
As an outcome of her work as an NCI/AcademyHealth Visiting Scholar, Dr. Virginia LeBaron, Kluge-Schakat Associate Professor of Compassionate Care at the University of Virginia School of Nursing, has a new paper accepted for publication in Oncology Nursing Forum, the research journal of the Oncology Nursing Society.
In collaboration with Dr. Sandra Mitchell and colleagues at NCI, the study examined end-of-life–related inquiries from patients with cancer and caregivers to NCI’s Cancer Information Service between 2018 and 2024. Findings highlight key information needs related to healthcare services, clinical trials, and palliative and hospice care, underscoring opportunities to improve patient- and caregiver-centered models of care at the end of life.
Citation:
LeBaron, V., Mitchell, S. A., Huang, G., Ng, D., de Moor, J. S., & Vanderpool, R. C. (2026). Exploring information needs of patients and caregivers experiencing cancer: A descriptive and network analysis of end-of-life inquiries to the National Cancer Institute's Cancer Information Service. Oncology Nursing Forum. (In press; accepted January 2026).
HDRP Fiscal Year (FY) 2025 Grant Portfolio Updates
In FY 2025, NCI funded 35 new grants totaling over $27 million, which are held by HDRP. These grants support research across the cancer continuum to improve cancer prevention, detection, diagnosis, treatment, and survivorship care.
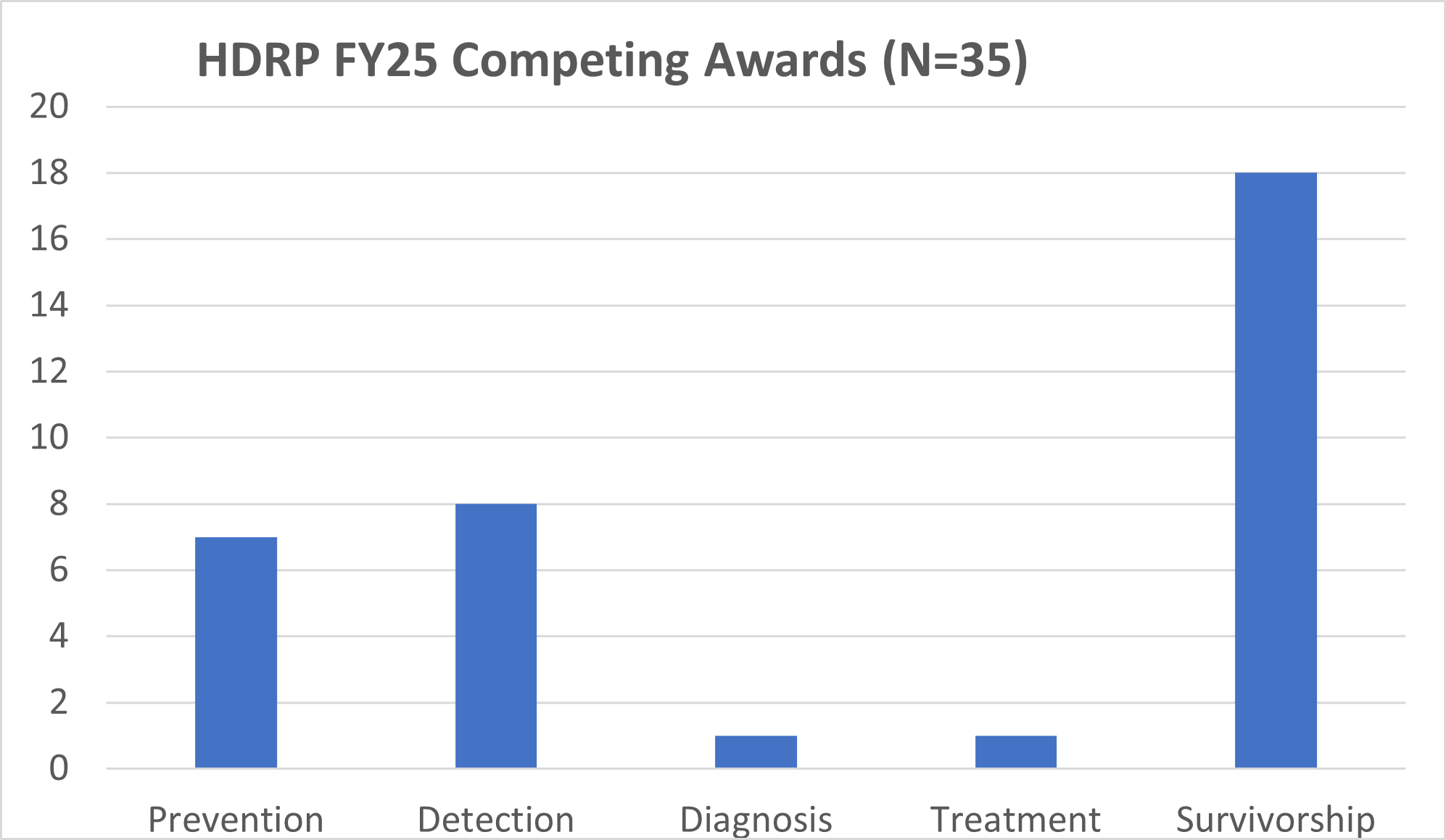
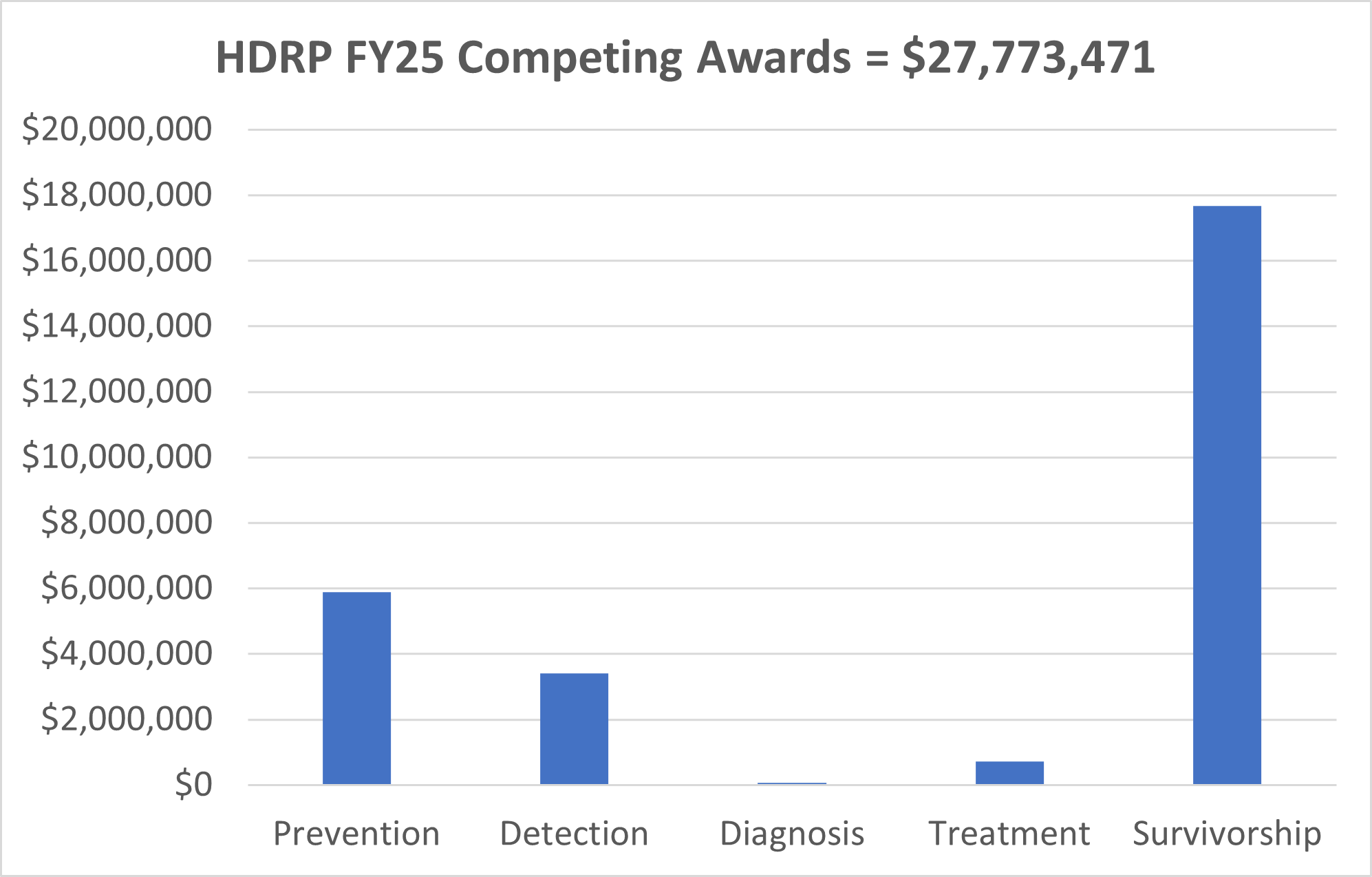
For a complete listing of all grants currently funded by NCI and held in HDRP, visit our funded grants page.
page.
Policy Updates
Grants-related Information
Grant-related policies continue to evolve in 2026. You can find notices of NIH policy changes here . Below are links to notices that may be most useful to you:
. Below are links to notices that may be most useful to you:
- NOT-CA-26-010
 - Notice of Extension of the Expiration Date for PAR-23-059 "National Cancer Institute Program Project Applications for the Years 2023, 2024, and 2025 (P01 Clinical Trial Optional)"
- Notice of Extension of the Expiration Date for PAR-23-059 "National Cancer Institute Program Project Applications for the Years 2023, 2024, and 2025 (P01 Clinical Trial Optional)" - NOT-CA-26-003
 - Notice of NCI's Participation in PA-25-302, NIH Small Research Grant Program (Parent R03 Clinical Trial Not Allowed)
- Notice of NCI's Participation in PA-25-302, NIH Small Research Grant Program (Parent R03 Clinical Trial Not Allowed) - NOT-OD-26-034
 - Guidance on Salary Limitation for Grants and Cooperative Agreements FY 2026
- Guidance on Salary Limitation for Grants and Cooperative Agreements FY 2026 - NOT-OD-26-033
 - Adjusted Timeline for NIHs Implementation of Common Forms
- Adjusted Timeline for NIHs Implementation of Common Forms - NOT-OD-26-026
 - NIH Requires Use of the eRA Prior Approval Module for the Submission of ALL Prior Approval Requests
- NIH Requires Use of the eRA Prior Approval Module for the Submission of ALL Prior Approval Requests - NOT-OD-26-019
 - Updated Application Policies: NIH Administrative Burden Reduction Effort Removal of Requirements for Letters of Intent and Unsolicited Applications Requesting $500,000 or More in Direct Costs
- Updated Application Policies: NIH Administrative Burden Reduction Effort Removal of Requirements for Letters of Intent and Unsolicited Applications Requesting $500,000 or More in Direct Costs
NIH News You Can Use!
This section of the HDRP newsletter highlights resources from the NIH Office of Extramural Research’s Extramural Nexus . It is the extramural scientific research community’s source for news on grants policies, processes, and more.
. It is the extramural scientific research community’s source for news on grants policies, processes, and more.
See below for some news that may be relevant to you!
- Planning an NIH Application involving Basic Experimental Studies with Humans (BESH)?

- NIH Launches New Central Resource to Support Replication and Reproducibility

- Aligning HIV/AIDS Receipt Dates with General Standard Receipt Dates to Simplify Application Processes and Reduce Burden for Researchers

- Check Out the Latest How to Apply – Application Guide

- How Can I Find Funding Opportunities Now That They Are Not in the NIH Guide?

- Announcing New Application and Award Structure for NIH-Funded International Collaborations (Replacing Foreign Subawards)

- Introducing Highlighted Topics: A Centralized and Simple Resource to Learn About Research Areas of Interest to NIH

- Updates to NIH Inclusion Policy

To subscribe to the blog, visit the NIH Extramural Nexus homepage .
.
Grantsmanship Resources
HDRP offers a “one-stop-shop” of relevant links to help investigators learn more about NIH and NCI information and policies related to writing and submitting new, resubmission, late, and renewal grant applications. Visit the Grantsmanship Resources page here. Please check with your program director to ensure information is current.
The NIH Grants and Funding site has been redesigned to address the needs of the extramural community. Visit the new grants and funding page .
.
HDRP Funding Opportunities
NIH continues to release highlighted topics for investigator-initiated grant application, representing priority areas within one or more NIH Institutes, Centers, or Offices (ICOs). NIH recently published Optimal Interprofessional Teaming and Care Coordination Strategies for Cancer Care Quality and Outcomes
for investigator-initiated grant application, representing priority areas within one or more NIH Institutes, Centers, or Offices (ICOs). NIH recently published Optimal Interprofessional Teaming and Care Coordination Strategies for Cancer Care Quality and Outcomes , which is being led by Drs. Sallie Weaver and Veronica Chollette.
, which is being led by Drs. Sallie Weaver and Veronica Chollette.
Below are notices of funding opportunities (NOFOs) that are sponsored or co-sponsored by HDRP. For the most up-to-date list, visit the HDRP funding opportunities webpage. For other NOFOs in cancer control, visit the DCCPS funding webpage .
.
| Announcement Title | Announcement Number | Expiration Date | Program Contact | NOFO Videos & Webinars |
|---|---|---|---|---|
| Cancer Prevention and Control Clinical Trials Grant Program (R01 Clinical Trial Required) | PAR-25-167 |
January 8, 2027 | Tanya Agurs-Collins 240-276-6956 collinsta@mail.nih.gov |
|
| Cancer Prevention and Control Clinical Trials Planning Grant Program (U34 Clinical Trials Optional) | PAR-25-103 |
October 26, 2027 | Rachelle Brick |
|
| Cancer Prevention and Control Clinical Trials Planning Grant Program (R34 Clinical Trials Optional) | PAR-25-104 |
October 26, 2027 | Stephanie Land |
|
| Dissemination and Implementation Research in Health (R21 Clinical Trial Optional) | PAR-25-143 |
January 8, 2028 | Gila Neta 240-276-6785 gila.neta@nih.gov |
|
| NIH Research Project Grant (Parent R01 Clinical Trial Not Allowed) | PA-25-301 |
January 8, 2028 | Research Program Contacts |
|
| Exploratory Grants in Cancer Control (R21 Clinical Trial Optional) | PA-25-253 |
September 8, 2028 | Mukesh Verma 240-276-6889 vermam@mail.nih.gov |
