HDRP Newsletter, February 2024
Message from the Acting Associate Director

On behalf of all my colleagues in the Healthcare Delivery Research Program (HDRP), I wish you a happy and healthy new year. I hope everyone enjoyed a joyous holiday and a restorative end to 2023. We look forward to continuing to work with you in 2024 to advance our shared goal of advancing innovative research to improve the delivery of cancer-related care.
In this first newsletter of 2024, we highlight several funding opportunities, recent and upcoming events, and multiple data resources. The newsletter also includes a snapshot of HDRP’s FY’23 grant portfolio and information about recent grants policy. We also hope you enjoy the “research spotlight” highlighting NCI’s investment in supporting research to improve Human Papillomavirus (HPV) Vaccine uptake to eliminate cervical cancer in the United States.
I welcome you to reach out to me or any of our staff to learn more about the work of HDRP and available funding opportunities and data resources for healthcare delivery research. A full staff listing is available on our HDRP Staff page.
Sincerely yours,
Janet de Moor, PhD, MPH
HDRP Events: Upcoming and Recap
HDRP continues to lead efforts that highlight scientific priorities for healthcare delivery research. Visit our News & Events page for a comprehensive listing of events. A recap of recent HDRP-led webinars and workshops is listed below.
SEER-MHOS Webinar Series
The National Cancer Institute (NCI) supports a research resource linking Surveillance Epidemiology and End Results (SEER) cancer registry data with Medicare Health Outcomes Survey (MHOS) data from the Centers for Medicare & Medicaid Services. SEER-MHOS offers investigators the opportunity to examine cancer health outcomes, such as health-related quality of life and activities of daily living, along with clinical and epidemiologic information. This webinar series was intended for researchers at all levels interested in discovering more about the SEER-MHOS data resource.
- Webinar 1, Propensity Scoring and Health Outcomes Data: Comparing Cancer and Non-Cancer Populations in the SEER-MHOS Data Resource (March 14, 2023)
- Webinar 2, Using Medicare Part D Claims in SEER-MHOS: A Guide for Researchers (June 20, 2023)
- Webinar 3, Overview of Two SEER/CMS Patient Survey Data Resources at NCI: A Fireside Chat for New Users (September 22, 2023)
Healthcare Teams 2023-2024 Cyber Discussion Series
The goal of this webinar series was to discuss lessons learned by researchers and cancer care delivery teams about how to continue cancer care and research operations and work effectively with key stakeholders during some of the nation's most catastrophic events. Care coordination needs among populations that experience cancer disparities (racial/ethnic minority, under-resourced and historically disadvantaged communities) and populations disproportionately affected by public health emergencies were a focus.
Three of the four sessions are available for viewing online. Below are additional details for where watch the webinars and how to join HDRP for the final session in January 2024:
- Session 1, Teaming, Coordination Challenges, and Approaches to Cancer Care During Disasters and Public Health emergencies (May 4, 2023)
- Session 2, Research Examining Disaster Impact & Resilience in Cancer Care Delivery (June 30, 2023)
- Session 3, Global Perspectives on Cancer Care Teaming and Resilience During Natural and Public Health Emergencies (October 10, 2023)
- Upcoming Session 4, Patient and Caregiver Experiences Navigating Cancer During Emergencies (March 7, 2024). In this workshop, participants will gain essential insights into managing cancer care during natural disasters and public health emergencies. Speakers will discuss emergency preparedness for patients and healthcare providers, accessing critical healthcare services, and fostering resilience in times of crisis. Discover practical strategies for continuity in cancer care and hear firsthand from clinicians, patients and caregivers who have successfully navigated cancer care during an emergency.
May 2023
- On May 15-16, 2023, HDRP hosted the HEROiC Conference on Health Economic Research Collaborations to Improve Cancer Care (a NCI Collaborative Cancer Health Economics Research Conference) which brought together stakeholders from government agencies and non-profit patient-advocacy, payer, and professional organizations engaged in cancer health economics research to identify areas of shared interest and synergy and to implement plans for collaborative activities to further this field. The Conference was organized as part of HEROiC, the Interagency Consortium to Promote Health Economics Research on Cancer.
June 2023
- On June 21, 2023, HDRP hosted a webinar entitled, Measuring Healthcare Organization Characteristics in Cancer Care Delivery Research. This webinar shared the results from an Agency for Healthcare Research and Quality (AHRQ) technical brief identifying:
- Gaps in evidence concerning how healthcare organizational characteristics impact cancer care and cancer-related outcomes, and
- Methodologic approaches and measurement tools needed to better understand the impact of organizational context and processes on the delivery of and outcomes associated with cancer screening, diagnosis, and treatment.
September 2023
- On September 26-27, 2023, HDRP hosted the second HEROiC Conference on Health Economic Research Collaborations to Improve Cancer Care Conference which involved stakeholders from non-profit cancer centers and academic institutions to further identify areas of shared research interest and collaboration. This meeting builds upon the May 2023 meeting to further identify and refine areas for potential collaboration across the entire cancer control continuum with input from this complementary group of stakeholders.
- On September 29, 2023, HDRP hosted Using Medicare Advantage Encounter Data in Cancer Research, a webinar that introduced researchers to the structure and content of Medicare Advantage Encounter data. Considerations and approaches for the use of these data when linked to the National Cancer Institute (NCI) Surveillance Epidemiology and End Results (SEER) cancer registry data were also discussed.
November 2023
- On November 16, 2023, HDRP hosted Measures and Methods to Advance the Science of Teaming and Coordination in Cancer Care. This virtual workshop that brought together cancer control, healthcare delivery, organizational behavior, and teamwork scientists to discuss measurement and methodological approaches that can strengthen the rigor and reproducibility of care delivery research on teamwork and care coordination across the cancer care continuum. Please visit this event page for detailed information about the meeting, including session videos and summaries.
Research Spotlight
Human Papillomavirus (HPV) Vaccine to Eliminate Cervical Cancer in the United States
Cervical cancer mortality is highest among those with insufficient access to healthcare. Routine screening is effective but must be repeated and is expensive. If 80% of eligible adolescents receive two doses of the human papillomavirus (HPV) vaccine, we could reduce the need for adult screening and still come close to eliminating death from cervical cancer.
Delivering the HPV vaccine has been challenging. In 2012, five years after the Centers for Disease Control and Prevention (CDC) recommended the vaccine, uptake was only 28% (girls only, at the time). The President’s Cancer Panel focused on this poor uptake; their report spurred NCI to action. Increasing uptake of HPV vaccine has been a substantial focus for DCCPS since. Collaborations with CDC, the American Cancer Society (ACS), and NCI-designated cancer centers have contributed to steady and significant improvement. Work remains to be done, but progress has been substantial. CDC data from 2022
focused on this poor uptake; their report spurred NCI to action. Increasing uptake of HPV vaccine has been a substantial focus for DCCPS since. Collaborations with CDC, the American Cancer Society (ACS), and NCI-designated cancer centers have contributed to steady and significant improvement. Work remains to be done, but progress has been substantial. CDC data from 2022 showed that 76% of boys and girls received ≥1 HPV vaccine dose and 62.6% were up to date with HPV vaccination.
showed that 76% of boys and girls received ≥1 HPV vaccine dose and 62.6% were up to date with HPV vaccination.
Tying HPV vaccine uptake to cervical cancer mortality reduction takes time; the vaccine is for adolescents before exposure and potential infection, and cancer develops slowly following persistent infection. However, U.S. data from CDC (Markowitz et al., 2016, Pediatrics ) show a 88%
) show a 88% decrease in HPV infection in girls aged 14-19 (considering only the types targeted by the vaccine). These data lead to great optimism that fewer and fewer American women will die from cervical cancer.
decrease in HPV infection in girls aged 14-19 (considering only the types targeted by the vaccine). These data lead to great optimism that fewer and fewer American women will die from cervical cancer.
- Administrative supplements were awarded to NCI-designated cancer centers in 2014, 2017, and 2020. Supplements supported research to characterize HPV vaccine uptake within the local community near each cancer center with the long-term goal of increasing HPV vaccine utilization and reducing HPV-related cancers. The three rounds of HPV vaccination supplements focused on leveraging local data, building local community collaborations, and addressing local sources of misinformation. This initiative formed an on-going coalition of cancer researchers focused on HPV vaccination.
- Grantees began holding open meetings, at their own initiative.
- The most recent annual meeting took place on February 17 and 23, 2023. The next annual meeting will be in April 2024 in Lexington, KY. For additional information and resources, visit our HPV Vaccine Uptake in Cancer Centers page.
- For the first time, NCI designated cancer centers collectively released a statement endorsing HPV vaccine uptake.
- Grantees began holding open meetings, at their own initiative.
- Over 400 applications for funding to study HPV vaccine uptake have been received by DCCPS, and more than 50 have been awarded.
- A PAR entitled, “Linking the Provider Recommendation to Adolescent HPV Vaccine Uptake” (issued in 2017) increased the average annual applications from 27 to 46 (through 2022).
- In collaboration with ACS and CDC, capacity-building efforts have continued:
- Hosted by CDC, NCI and extramural experts, state cancer and immunization programs were selected for technical assistance meetings in 2018 and 2019. For these meetings, technical assistance was provided in HPV vaccination uptake interventions, measuring HPV vaccinations at clinic and community levels, and where to find expert assistance as the projects continued. These meetings focused on promoting local communication and goal-setting.
- There have been annual National HPV Vaccine Roundtable Meetings, with NCI and CDC participation as well as over 70 member organizations across the United States.
HDRP Fiscal Year (FY) 2023 Grant Portfolio Updates
In FY 2023, NCI funded 53 new grants totaling over $28 million, which are held by HDRP. These grants support research across the cancer continuum to improve cancer prevention, detection, diagnosis, treatment, and survivorship care.
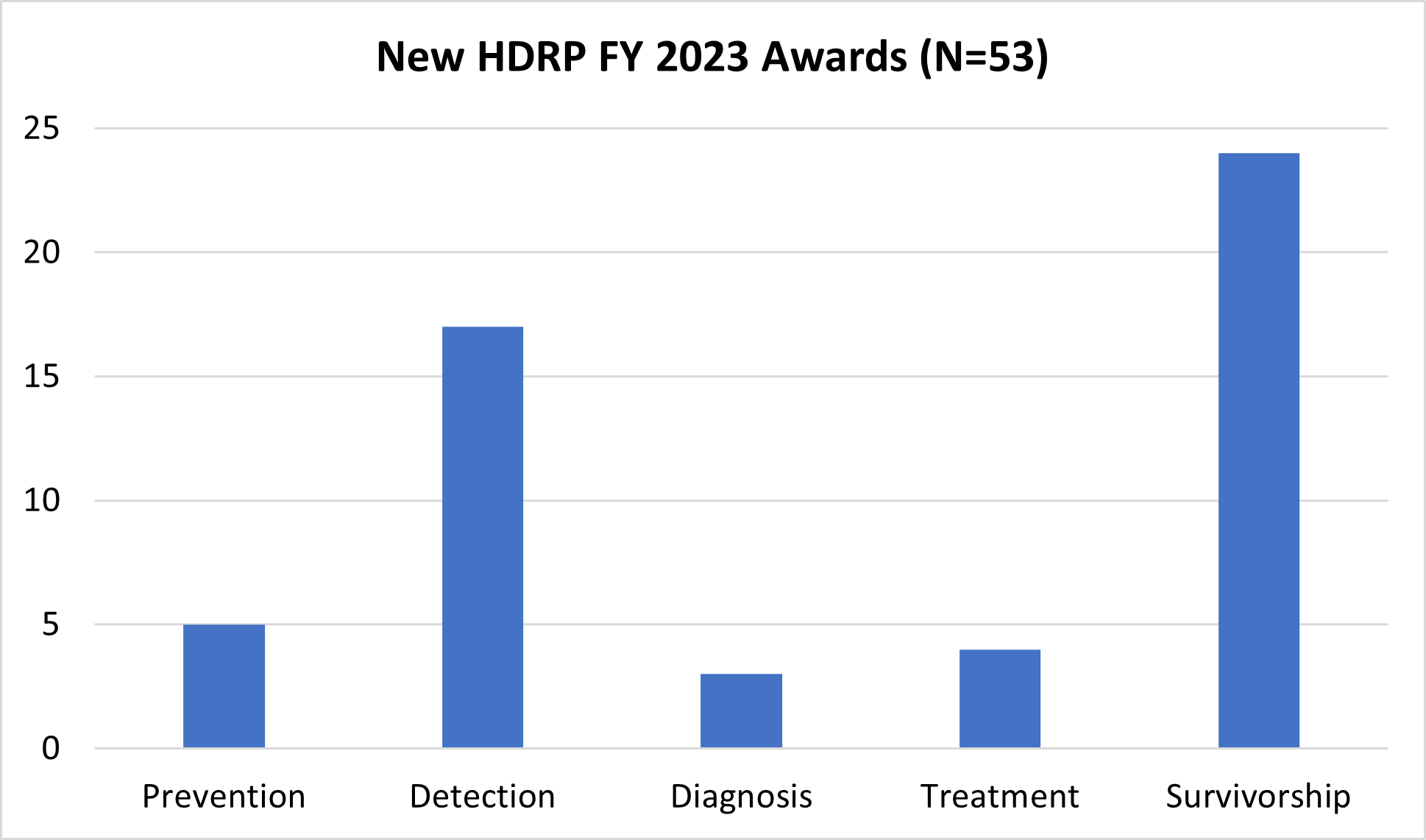
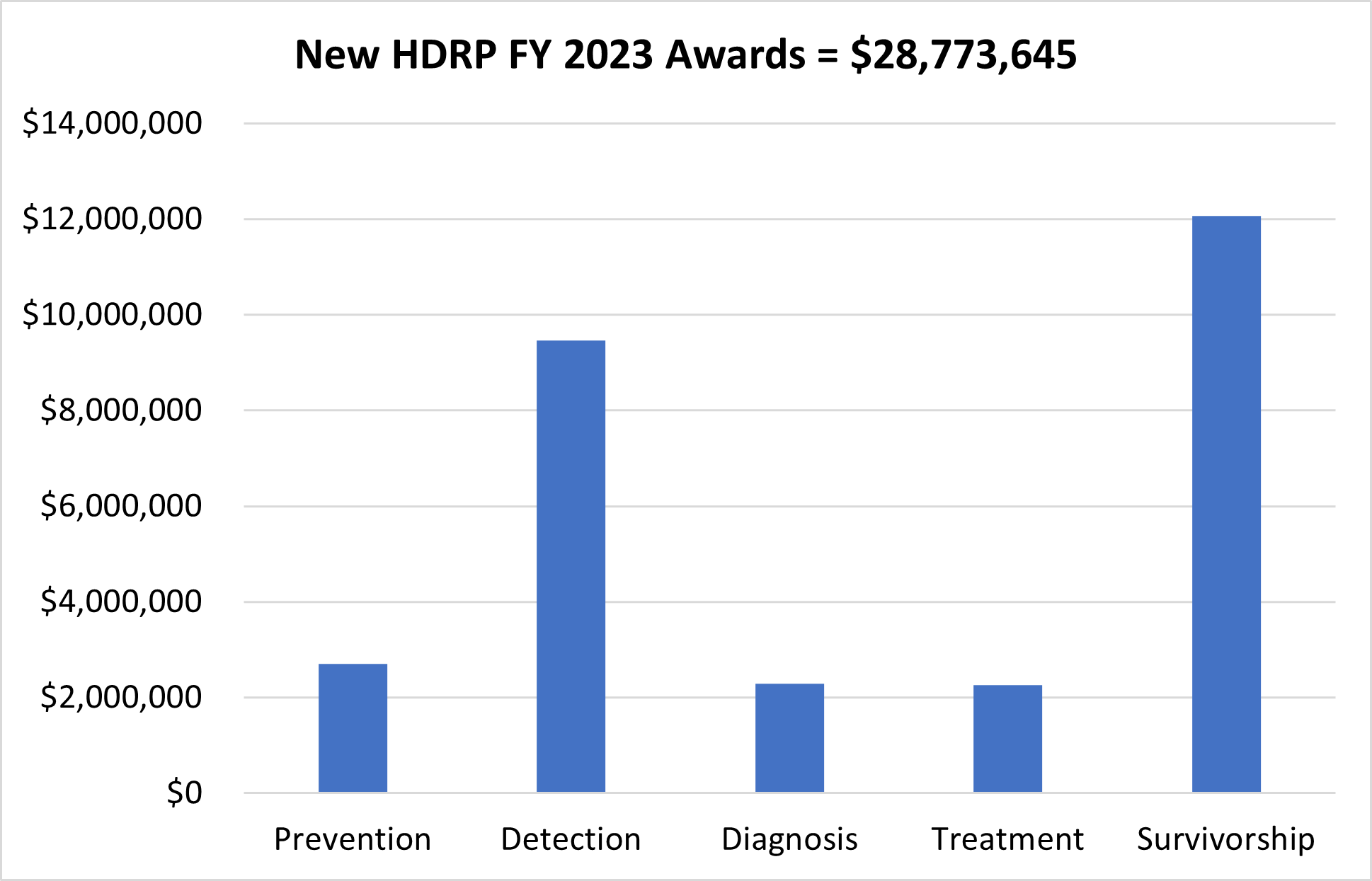
For a complete listing of all grants currently funded by HDRP, visit our funded grants page.
page.
Policy Updates
Grants-related Information
Funding Policy Updates
The NCI is currently operating under an FY 2024 continuing resolution that funds the government at FY 2023 levels. As a result, we are currently functioning on interim paylines. Visit the Division of Extramural Activities website for information on the Institute's budget, funding strategy, and more. The table below provides a snapshot of different funding mechanisms and associated paylines.
website for information on the Institute's budget, funding strategy, and more. The table below provides a snapshot of different funding mechanisms and associated paylines.
| Mechanism | FY 2024 Funding Policy |
|---|---|
| Experienced and New Investigator R01 | 9th Percentile |
| Early-stage Investigator R01 | 14th Percentile |
| Exploratory/Developmental (R21) | 9th Percentile |
| Small Grant (R03) | 25 Impact Score |
AREA Grant (R15) Grant (R15) |
25 Impact Score |
Also, visit the NIH Grants & Funding page for more information on Standard Application Due Dates .
.
NIH Policy for Data Management and Sharing (DMS)
On January 25, 2023, the final NIH DMS Policy came into effect for any NIH-funded research that will generate scientific data, regardless of the research grant budget size. As a refresher, HDRP has compiled information from NIH on preparing DMS plans, research subject to the new NIH DMS Policy, and requesting and justifying costs for DMS. Information is available here:
came into effect for any NIH-funded research that will generate scientific data, regardless of the research grant budget size. As a refresher, HDRP has compiled information from NIH on preparing DMS plans, research subject to the new NIH DMS Policy, and requesting and justifying costs for DMS. Information is available here:
Working closely with the NIH, the Federal Demonstration Partnership (FDP) – an association of federal agencies, research policy organizations, and academic research institutions – invites feedback from researchers by participating in a pilot of DMS plan templates. For more information, visit the NIH’s Extramural Nexus post
(FDP) – an association of federal agencies, research policy organizations, and academic research institutions – invites feedback from researchers by participating in a pilot of DMS plan templates. For more information, visit the NIH’s Extramural Nexus post and FDP website.
and FDP website.
Announcements
NCI’s Specialized Programs of Research Excellence (SPOREs) – U54
On November 24, 2023, the NCI released a notice of intent to publish a notice of funding opportunity (NOFO) for a Specialized Programs of Research Excellence (SPOREs) in cancer health disparities and minority health. This program will support a network of multidisciplinary, multi-institutional U54 SPOREs – multi-project awards focusing uniquely on health disparities and/or minority health translational research to improve cancer care for underserved populations.
The goals of the proposed research may vary widely and addresses for example:
- Discovery, validation, and assessment of how various determinants of health intersect with the biology of cancer to affect cancer incidence, diagnosis, treatment, early detection, and prevention in populations who are underserved.
- Characterization of the biological impact of social determinants of health and identifying specific biological pathways that might be targeted by clinical or public health interventions.
- Hypothesis generating studies characterizing risk factor prevalence or biological differences in populations who are underserved.
- Investigating the role of determinants of health and comorbidities on toxicity to therapeutic interventions in populations who are underserved.
Visit the website (NOT-CA-24-008 ) for more information on this highly competitive NOFO. The due date for applications is September 25, 2024.
) for more information on this highly competitive NOFO. The due date for applications is September 25, 2024.
HDRP Funding Opportunities
This section includes links to new highlighted NOFOs on healthcare delivery research published since the last edition of this newsletter. For a complete list of NOFOs, visit our funding opportunities webpage.
| Announcement Title | Announcement Number | Expiration Date |
|---|---|---|
| NOSI: Addressing Cancer-Related Financial Hardship to Improve Patient Outcomes | NOT-CA-22-045 | March 1, 2024 |
| NOSI: Telehealth in Cancer Care | NOT-CA-21-043 | March 8, 2024 |
| NOSI: New Information Technology-Enabled Care Delivery Models to Improve Depression Care in Cancer | NOT-CA-21-085 | March 8, 2024 |
| Resource Center for Cannabis and Cannabinoid Research (U24 Clinical Trial Not Allowed) | RFA-AT-24-006 | April 17, 2024 |
| Coordinating Center to Support Multi-Sectoral Preventive Interventions that Address Social Determinants of Health in Populations that Experience Health Disparities (U24, Clinical Trial Not Allowed) | RFA-OD-24-006 | August 6, 2024 |
| Cancer Prevention and Control Clinical Trials Planning Grant Program (R34 Clinical Trials Optional) | PAR-22-173 | September 8, 2025 |
| Cancer Prevention and Control Clinical Trials Planning Grant Program (U34 Clinical Trials Optional) | PAR-22-174 | September 8, 2025 |
Data & Other Resources
SEER-CAHPS Data Available!
SEER-CAHPS is a publicly accessible data resource developed to enable investigators to conduct research on patient-reported experiences and quality of cancer care. These data provide a rich opportunity for analyses of Medicare beneficiaries’ experiences with their care at various stages of the cancer care continuum.
The SEER-CAHPS data resource was last updated in 2023 and includes the following years of data:
- SEER Cancer Registry Data Through 2019
- CAHPS Experience of Care Survey Data Through 2019
- Medicare Claims Data Through 2019
Updated SEER-MHOS Data Available!
The SEER-MHOS is a data resource for cancer health outcomes research. It is based on a linkage between SEER cancer registry data and the CMS Health Outcomes Survey .
.
The SEER-MHOS team is excited to announce that the most recent linkage was completed in 2023. It contains data from 22 SEER registries. The data resource currently spans from 1998 through 2021. Over 900,000 survey respondents without cancer in the same geographic areas are included as a non-cancer comparison group.
Updated SEER-Medicare Data Available!
SEER-Medicare data reflects the linkage of two large population-based sources of data that provide detailed information about Medicare beneficiaries with cancer.
The SEER-Medicare team has recently updated the years of data in this data resource to include all Medicare eligible persons appearing in the SEER data who were diagnosed with cancer through 2019, and their Medicare claims through 2020.
For additional information on SEER-linked data and other tools and resources, visit our Data & Tools page.
HDRP News You Can Use!
Employment Opportunities
Chief, Healthcare Assessment Research Branch (HARB)
NCI welcomes letters of interest to serve as Chief of the Healthcare Assessment Research Branch (HARB) within the Division of Cancer Control and Population Sciences (DCCPS). Please visit our HARB webpage for more information on HARB’s mission and vision.
Letters of interest and CVs are encouraged from clinical, health services, and public health researchers with a strong record of scholarship in relevant areas, supervisory experience, and familiarity with the extramural community. Materials should be submitted to NCIHDRP@mail.nih.gov by April 1, 2024, with “HARB Chief” as the email subject line.
Cancer Research Training Award (CRTA): Healthcare Assessment Research Branch (HARB)
The CRTA Fellow would work on projects within HARB that are directly related to the branch’s research priorities, including enhancements to SEER-Medicare and SEER-Medicaid, developing novel linkages between SEER and other sources of data, and more. The future Fellow would have opportunities to collaborate on manuscripts, lead their own research projects, as well as support other branch research initiatives as needed.
The branch welcomes interested candidates for this full-time, paid post-doctoral fellowship based in Rockville, MD.
Training Opportunities
The training section of our website provides information on NIH/NCI-relevant opportunities. Visit to learn more about the Cancer Prevention Fellowship Program, the Presidential Management Fellows Program, as well as the Health Communications Internship Program.
